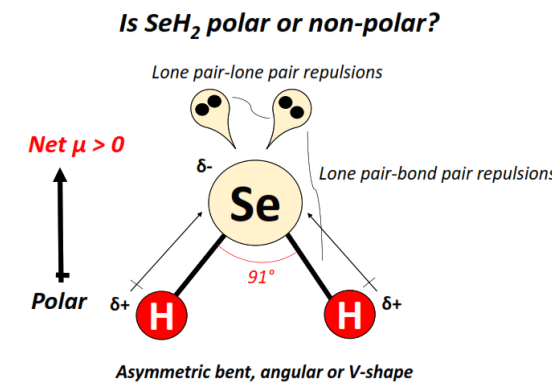
C2H6O Lewis Structure (Dimethyl Ether) | C2H6O Lewis Structure (Dimethyl Ether) Several people get confused when they look at the chemical formula of C2H6O as it is a general formula for both...

Is n pentane an ionic molecular nonpolar or molecular polar compound what intermolecular forces are present? - Opera Residences

SO3 Lewis structure, molecular geometry or shape, electron geometry, bond angle, formal charge, | Molecular geometry, Molecular, Pi bond

The two isomers having the formula C2H6O boil at -23 degC and 78.5 degC. Draw the structure of the isomer that boils at -23 degC and of the isomer that boils at
SOLVED: A liquid that would be soluble in water would be? Select one: a. benzene; C6H6 b. pentane C5H12 c. dichloromethane CH2Cl2 d. methylbenzene C6H5CH3 e: ethanol C2H6O

Is CH3NH2 polar or non-polar? - Polarity of CH3NH2 | Molecular geometry, Covalent bonding, Electron configuration
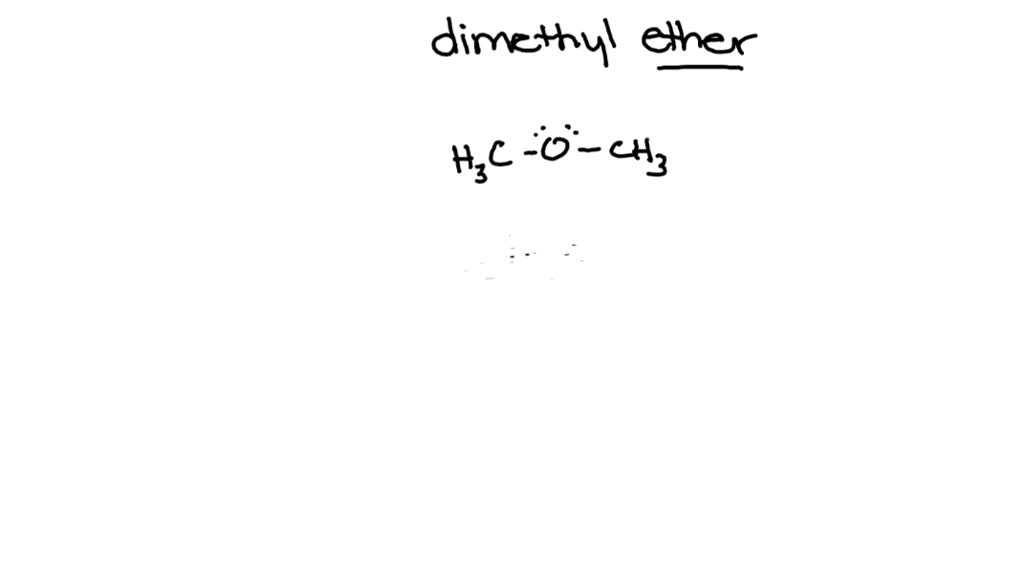
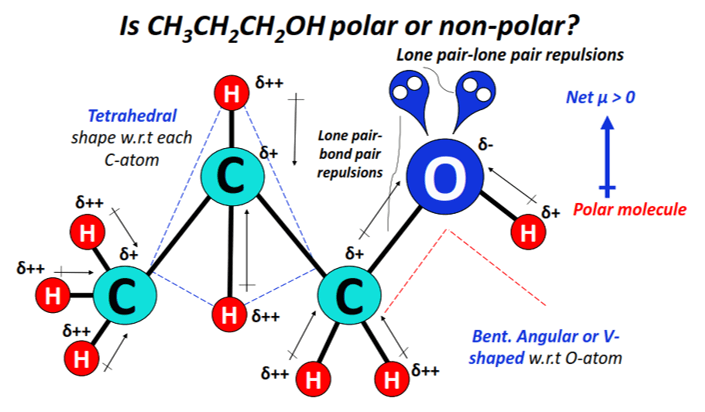

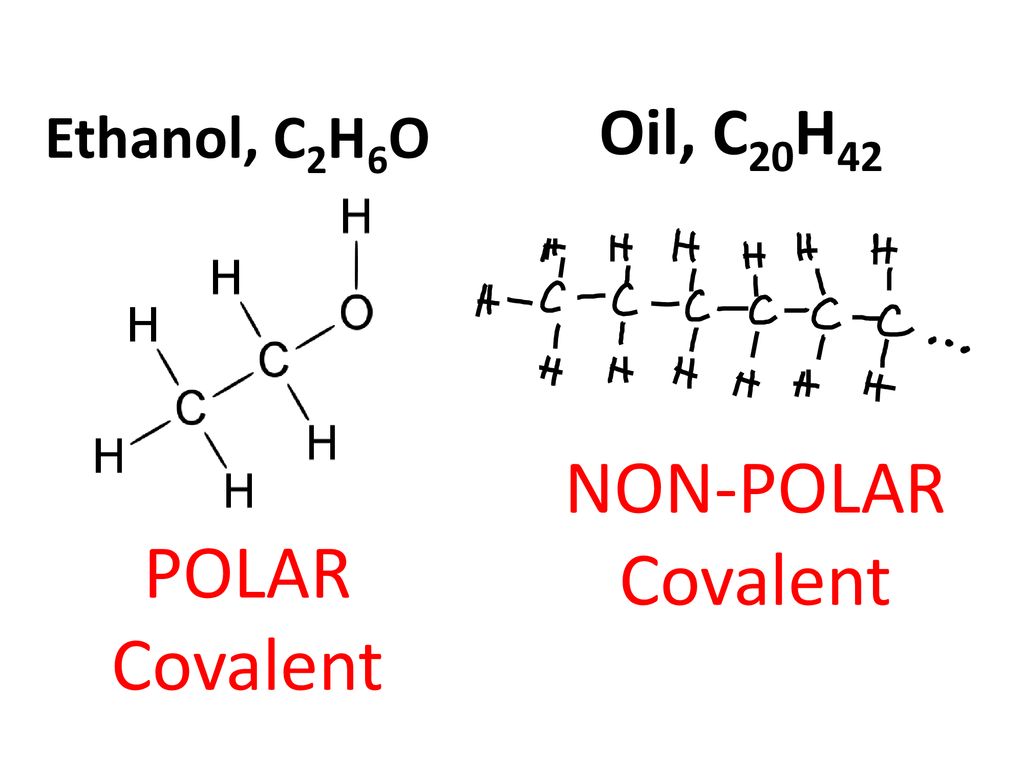






![Biology] Polar Bonds - General Discussion - Neowin Biology] Polar Bonds - General Discussion - Neowin](https://cdn.neowin.com/forum/uploads/post-107847-1188869267_thumb.jpg)