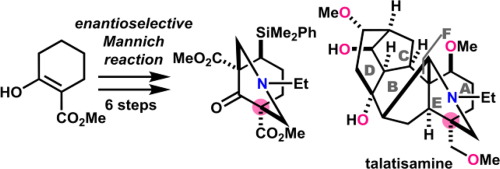
SOLVED: The following sequence of reactions was employed during synthetic studies on reidispongiolide A, a cytotoxic marine natural product (Tetrahedron Lett. 2009, 50, 5012-5014). Draw the structures of compounds A, B, C,

PDF) Stereoarrayed CF 3 -Substituted 1,3-Diols by Dynamic Kinetic Resolution: Ruthenium(II)-Catalyzed Asymmetric Transfer Hydrogenation

Tetrahedron Letters | Vol 43, Issue 12, Pages 2127-2321 (18 March 2002) | ScienceDirect.com by Elsevier

SOLVED: Draw the final products for the following reactions shown below and explain your answers to get full credits. CHO TFA . TF1,0 Am Chem Soc 2001 123,4370 POCl3; DMF Tetrahedron Lett.

Tetrahedron Letters | Vol 43, Issue 27, Pages 4717-4893 (1 July 2002) | ScienceDirect.com by Elsevier

Tetrahedron Letters | Vol 43, Issue 27, Pages 4717-4893 (1 July 2002) | ScienceDirect.com by Elsevier

Tetrahedron Letters | Vol 43, Issue 4, Pages 541-733 (21 January 2002) | ScienceDirect.com by Elsevier
Arylpalladium Phosphonate Complexes as Reactive Intermediates in Phosphorus-Carbon Bond Forming Reactions - UNT Digital Library

Highly Efficient Chemoselective Deprotection of O, O-Acetals and O, O-Ketals Catalyzed by Molecular Iodine in Acetone | PDF | Iodine | Ketone

Tetrahedron Letters | Vol 43, Issue 42, Pages 7451-7641 (14 October 2002) | ScienceDirect.com by Elsevier

Amphidinolide A Isolation: 1. Kobayashi, J. et al. Tetrahedron Lett. 1986, 27, Kobayashi, J. et al. J. Nat. Prod. 1991, 54, Biological Activity: - ppt download